Patient model systems of neurodegenerative diseases
Neuroscience research presents a particular challenge. You can’t study brain cells taken from living patients. Our researchers are developing models of cells in the nervous system using skin cells. And they’re using these models to find new potential treatments.

Reprogramming cells
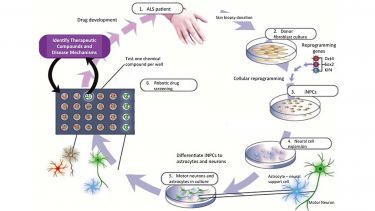
How do you study the cells affected by neurodegenerative diseases when you can’t use samples taken from living patients? This is a question asked by Dr Laura Ferraiuolo’s research team at the Sheffield Institute for Translational Neuroscience (SITraN). And it's an important one if we want to develop viable treatment options for patients.
Laura and colleagues have developed a unique way to reprogramme skin cells into neurons and glia. These innovative patient cell models contain all the genetic variation that is found between people with neurodegenerative disease, their family members and control volunteers.
Importantly, the cell models also uniquely retain features of ageing. This is important to the onset and progression of neurodegenerative conditions. The models provide a powerful way to study what is going wrong in the cells that degenerate. Which could help us to uncover new therapeutic targets.
Cell models are being used to understand the causes of disease. Researchers are then undertaking rapid, high-volume drug screens to find therapies that are targeted at different genetic causes and disease mechanisms.
We have developed human cellular models of neurons and the support cells of the nervous system called glia. These models came from small skin biopsies donated by patients and healthy control volunteers in order to study diseases like motor neuron disease (MND), Parkinson’s and Alzheimer’s disease. We have learnt that diseases like MND really consist of a number of different subtypes. Effective treatments are more likely to be found if we can better identify an MND sub-type and have a more personalised approach to treatment.
Dr Laura Ferraiuolo
Lecturer in Translational Neurobiology
AMBRoSIA
Our researchers have collected hundreds of patient biosamples through an MNDA-funded programme called AMBRoSIA. They use these to create neuron and glia patient model systems to identify subtypes for MND patients where there is no clear genetic cause. A similar collection of cells is available to study Alzheimer’s and Parkinson’s disease
The team have attracted a number of industry collaborations. BenevolentAI, Quell Therapeutics, Pfizer, and AstraZeneca are just some of the companies that they are working with to unravel the biological mechanisms of neurodegeneration and find new therapies.
The use of these cells has led to findings that have supported clinical trials. UDCA for Parkinson’s disease is currently in a trial with patients, and the cells supported pre-clinical tests for upcoming clinical trials in MND in collaboration with AclipseOne.
In addition, the unique ability of these reprogrammed cells to model ageing will help us unravel the physiological mechanisms of ageing, as well as neurodegeneration, to promote healthy ageing.
We are aiming towards being able to offer patients a tailored personalised medicine approach with targeted therapeutics for diseases with currently few or ineffective treatment options.