Lithium-ion batteries have undoubtedly powered the last 20 years of technological advancement, from the ubiquity of smartphone technology to the growing fleet of <1 million battery-powered vehicles currently on the roads worldwide.
There are, however, growing concerns over the sustainability of lithium production, due to problems such as the chemical contamination of water supplies around lithium mines, the wealth distribution of the proceeds of lithium mining and the environmental impact of disposing of the spent batteries.
Finding an alternative technology is, therefore, a top priority for Government science and technology funding.
One such notable technology is sodium-based batteries. Dr Nik Reeves-McLaren, Senior Lecturer in Energy Materials at the University of Sheffield, has received funding from the Faraday Institution as part of the NEXGENNA project, to research these sodium-based energy storage materials to power a greener future.
There is a wide range of literature that suggests sodium has the ability to perform well in a battery, especially when the energy density is less important than the cost per watt.
Led by Prof. John Irvine at the University of St Andrews - in collaboration with Royce Partners, the Universities of Sheffield and Cambridge - NEXGENNA will accelerate the development of sodium-ion battery technology.
Through development of safe sodium-ion batteries with high performance and long life cycles, the project will bring down the cost of electric vehicles.
Dr. Nik Reeves-McLaren, an investigator on the NEXGENNA project said:
"The processes that occur when we remove large Na+ ions from sodium-ion battery electrode materials tend to be more complex, and occur over a greater number of voltage steps, than in their lithium-based cousins.
This isn't ideal for applications, and tends to have been poorly understood.
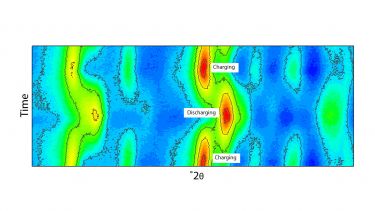
The Empyrean opens a new window on these important processes, and will allow us to much more rapidly optimise the performance of the next generation of cathode and anode materials for sodium-ion applications."
EQUIPMENT
The Malvern Panalytical Empyrean, available through Royce at the University of Sheffield, is capable of performing X-ray Diffraction during the charging and discharging cycles of sealed batteries. It uses a high-energy, short-wavelength silver (Ag) X-Ray tube which is capable of penetrating standard pouch cells and windowed coin cells.
Additionally, the diffractometer can be used to get detailed information not only on the crystallographic nature of the electrode, but also on the disorder in the material, known as the pair distribution function [PDF].
VISION
By the end of 2023, the NEXGENNA project aims to:
- Discover and develop innovative electrode materials for higher performance, lower cost Na-ion batteries.
- Discover and develop next-generation electrolyte materials, giving higher sodium mobility and therefore higher power.
- Refine the test and characterisation methods most applicable for materials for Na-ion batteries.
IMPACT
Utilising abundant minerals in energy storage materials will have a positive impact on the local environment by reducing the amount of toxic material leaching into the watercourses (both in their production and end of life); reduce global reliance on fossil fuels; and power the reusable energy revolution needed to limit the effects of man-made climate change.